Oncology
Abstract
Cancer-induced bone pain (CIBP) is among the most common and debilitating symptoms in patients with bone metastasis. Current treatments are somewhat effective but have severe side effects. For the future development of safer CIBP treatment, in this study, we sought to investigate the mechanisms whereby the cancer/nerve interaction controls CIBP. We found that c-Kit, a receptor tyrosine kinase, was activated in the dorsal root ganglia (DRG) sensory neurons of mice with CIBP and that c-Kit’s sole ligand, stem cell factor (SCF), was enhanced in the bone marrow with bone metastasis. When DRGs were treated SCF or conditioned medium from high SCF-expressing cancer cells, in vitro nerve sprouting was enhanced, and this effect was abolished with c-Kit inhibitors. Mice, intrafemorally inoculated with cancer cells that had varying SCF-expression developed CIBP and enhanced peripheral nerve sprouting in an SCF-dependent manner. Downstream proteomic analysis revealed that SCF upregulated and activated fibroblast growth factor 1 (FGF1) in DRGs. When FGF1 was knocked down in DRGs, SCF-mediated nerve sprouting was prevented. Taken together, our studies demonstrate the importance of the SCF/c-Kit axis in CIBP and nerve sprouting, and identify the SCF/c-Kit/FGF1 pathway as a potential therapeutic target for CIBP.
Authors
Kelly F. Contino, Jenna Ollodart, Yang Yu, Sun H. Park, Shunsuke Tsuzuki, Kara Rollins, Tyler M. Heethouse, Joshua Chu, Laiton R. Steele, Takahiro Kimura, Jingyun Lee, Cristina M. Furdui, Lance D. Miller, Fang-Chi Hsu, Yusuke Shiozawa
Abstract
Authors
Natalie E. Andresen, Heehwa G. Son, Joongho J. Joh, Shadmehr Demehri
Development and characterization of triazole-based WDR5 inhibitors for the treatment of glioblastoma
Abstract
Glioblastoma (GBM) cancer stem cells (CSCs) contribute to tumor recurrence, treatment resistance, and dismal clinical outcomes. Genetic and pharmacological evidence suggests that the nuclear scaffolding protein WD-repeat containing protein 5 (WDR5) is a therapeutic vulnerability of the CSC population. However, previously reported WDR5 inhibitors display low permeability and are unable to penetrate the blood-brain barrier (BBB), limiting their utility in GBM. Herein, we report the structure-guided development of a novel series of triazole-based WDR5 WIN-site inhibitors designed to increase passive brain penetration. We identified triazole-based WDR5 inhibitors that are potent, passively permeable, and in some cases more brain penetrant than other scaffolds. We phenotypically assessed our novel WDR5 inhibitors in a panel of patient-derived CSC models and uncovered unique WDR5-regulated metabolic genes in GBM. We also evaluated their antiproliferative activity against CSCs both in vitro and in vivo. Finally, to identify novel combination opportunities, we screened a 2,100-compound chemical probe library and identified that the ATAD2 inhibitor BAY-850 synergizes with WDR5 inhibitors to enhance CSC killing. Our work diversifies the chemical matter targeting WDR5, clarifies the in vitro consequences of WIN-site inhibition in CSCs, and encourages the future development of next-generation WDR5 inhibitors with the potential to achieve in vivo efficacy in the brain.
Authors
Jesse A. Coker, Steven R. Martinez, Sang Hoon Han, Anthony R. Sloan, Amit Kumar Gupta, George Bukenya, Paul Polzer, James H. Ramos, Emma G. Rico, Annabella Rico, A. Abigail Lindsey, Tanvi Navadgi, Natalie Reitz, Todd Romigh, Jonathan Macdonald, Dhiraj Sonawane, Christopher M. Goins, Christopher G. Hubert, Nancy S. Wang, Feixiong Cheng, Joseph Alvarado, Samuel A. Sprowls, Justin D. Lathia, Shaun R Stauffer
Abstract
BACKGROUND. Chimeric antigen receptor T-cell (CAR-T) therapies have revolutionized treatment for relapsed/refractory multiple myeloma (RRMM). However, cytokine release syndrome (CRS), a common and potentially severe complication, requires inpatient monitoring, limiting access and increasing costs. Wearable devices could support outpatient CAR-T delivery, but feasibility for CRS detection versus standard care remains unproven. METHOD. We conducted a prospective, single-center observational pilot study to assess the feasibility of using wearable devices for monitoring vital signs and detecting CRS. Thirty patients receiving idecabtagene vicleucel (ide-cel) or ciltacabtagene autoleucel (cilta-cel) were enrolled; 25 with sufficient monitoring data were evaluable. Sensors collected skin and axillary temperature, oxygen saturation, respiratory and heart rate, and motion. Peripheral blood cytokines were analyzed pre- and post-infusion using a multiplex proteomic platform. The primary outcome was feasibility, assessed by CRS detection sensitivity and specificity; secondary outcomes included adherence, lead time, and performance of models integrating wearable and cytokine data. RESULTS. CRS occurred in 20 of 25 patients. The best-performing wearable model detected 18 or 20 CRS episodes with a sensitivity of 0.72 (mean 0.75; 95% CI 0.60–0.91) and a specificity of 0.80 (mean 0.76; 95% CI 0.68–0.84), and a median lead time of 7:00 hours before nursing recognition. Median adherence during high-risk periods was 71%. Cytokine changes paralleled temperature elevations, and IFN-γ emerged as a consistent biomarker. CONCLUSION. Wearable devices are feasible for early CRS detection and may support outpatient CAR-T care. Larger outpatient studies are warranted. TRIAL REGISTRATION. This study did not meet the criteria for ClinicalTrials.gov registration.
Authors
Sridevi Rajeeve, Matt Wilkes, Nicole Zahradka, Lewis Tomalin, Mujahid Quidwai, Darren Pan, Nicholas J. Calafat, Martin Cusack, Adolfo Aleman, Kseniya Serebryakova, Katerina Kappes, Hayley Jackson, Sarita Agte, Santiago Thibaud, Larysa Sanchez, Shambavi Richard, Joshua Richter, Cesar Rodriguez, Hearn Jay Cho, Ajai Chari, Sundar Jagannath, Alessandro Laganà, Adriana C. Rossi, Samir Parekh
Abstract
BACKGROUND. Asparaginase is essential for curing acute lymphoblastic leukemia (ALL), but its use is limited by asparaginase-associated pancreatitis (AAP), a severe and unpredictable toxicity lacking validated prospective biomarkers. We sought to define early systemic molecular features of susceptibility to AAP. METHODS. We performed longitudinal lipidomic and proteomic profiling in two independent pediatric ALL cohorts (n = 161; 79 AAP cases, 82 controls) using paired blood samples collected before asparaginase exposure and at the end of induction therapy (including a single dose of asparaginase), thereby capturing pre-injury biology rather than consequences of pancreatitis. We applied differential abundance and network-based analyses, and integrated lipid–cytokine associations using proteomics. RESULTS. Across cohorts, we identified a reproducible lysophosphatidylcholine (LPC)–centered signature characterized by attenuated induction therapy-associated LPC responses and disruption of LPC co-regulation at the network level. Proteomic profiling revealed enrichment of cytokine signaling pathways, and integrative analyses demonstrated altered lipid–cytokine coupling, including a flip in association direction for LPC species and interleukin-18 (IL-18) between cases and controls. Although IL-18/LPC ratios do not differ globally, elevated post-induction IL-18/LPC ratios identify AAP risk within a protocol-defined very high-risk ALL subgroup (AUC = 0.81). CONCLUSION. These findings support a systems-level model in which failure of coordinated lipid–immune responses under therapeutic stress confers vulnerability to AAP, providing a framework for validation and mitigation strategies. TRIAL REGISTRATION. NCT00400946; NCT01574274; NCT03020030 (parent trials). FUNDING. Servier Pharmaceuticals (IIT-95014-027-USA); SDRC (P30DK116074); Stanford SPARK; Fonds de Recherche du Québec – Santé; Fondation Charles-Bruneau; The Leukemia & Lymphoma Society of Canada.
Authors
Cheng-Yu Tsai, Na Bo, Thai Hoa Tran, Maisam Abu-El-Haija, Gayathri Swaminathan, Bomi Lee, Sudhir Ghandikota, Li Wen, Yves Théorêt, Steven D. Mittelman, Elena J. Ladas, Anil G. Jegga, Lewis B. Silverman, Ying Ding, Sohail Z. Husain
Abstract
Immune checkpoint inhibitors have transformed cancer therapy, yet many patients fail to achieve durable responses due to insufficient T cell reinvigoration. Cytokines offer promise for enhancing immunotherapy, but their clinical use is limited by toxicity and a narrow therapeutic index. Immunocytokines, engineered fusion proteins combining antibody specificity with cytokine activity, aim to overcome these challenges by targeting cytokine delivery to immune cells or the tumor microenvironment. We describe SAR445877 (SAR’877), a novel PD-1-targeted immunocytokine that fuses a high-affinity anti-PD-1 antibody with a detuned IL-15/IL-15Rα sushi domain complex. SAR’877 blocks PD-1/PD-L1 and PD-1/PD-L2 interactions while selectively delivering IL-15 signals to PD-1+ T cells, enhancing proliferation and activation of antigen-experienced CD8+ and CD4+ T cells and NK cells, while minimizing systemic inflammation. Mechanistically, SAR’877 activates STAT5 signaling in PD-1+ lymphocytes and restores effector function in exhausted T cells. In preclinical models, a murine surrogate of SAR’877 accelerated viral clearance and induced robust anti-tumor immunity by expanding cytotoxic CD8+ T cells and promoting Th1 polarization. Notably, SAR’877 outperformed anti-PD-1 plus untargeted IL-15, highlighting the therapeutic potential of targeted IL-15 delivery. These findings position SAR’877 as a promising next-generation immunotherapy with enhanced efficacy and reduced cytokine-associated toxicities.
Authors
Isaraphorn Pratumchai, Marie Bernardo, Julien Tessier, Jaroslav Zak, Kristi L. Marquardt, Joon Sang Lee, Maheeka Bimal, AHyun Choi, Anthony M. Byers, Mikielia G. Devonish, Roberto Carrio, Dan Lu, Stella A. Martomo, Jeegar Patel, Yu-an Zhang, Ingeborg M. Langohr, Virna Cortez-Retamozo, Dinesh S. Bangari, Angela Hadjipanayis, Xiangming Li, Valeria R. Fantin, Donald R. Shaffer, John R. Teijaro
Abstract
Tumor-infiltrating CD8 cells recognize neoantigens created by tumor-specific mutations. Nonetheless, even after checkpoint inhibitor therapy, most patients progress. A deeper understanding of anti-tumor responses could facilitate development of better therapies. To enable such studies, we applied TCXpress, a high throughput platform that clones fully expressible TCRs from single cells into retro- or lenti- viral vectors without sequencing or gene synthesis, to study TCRs from CD8 cells infiltrating mouse MC38 tumors. We expressed cloned TCRs in reporter cells and interrogated TCR specificity by coculturing them with B6WT3 cells transduced with tandem minigenes encoding predicted neoantigens. We isolated TCRs reactive against epitopes from mutant Rpl18, Adpgk, Psmd2, and Zc3h7b along with self-reactive TCRs that recognized normal B6 and MC38 cells. Importantly, we successfully treated MC38-bearing mice with T cells transduced with anti-Rpl18 TCRs. These results establish a system that could be used to study many types of T cell responses and validates a therapeutic approach that could be tested in the clinic.
Authors
Alexander M. Rowe, Smriti Chaurasia, Wenzhong Wei, Laura García-Diéguez, Katherine Querry, Johnathon G. Schiebel, Christy Smolak, Alexander G. Muralles, Daniel Wikenheiser, Kevin Quann, Collin Pirner, Kentin Codispot, Mark J. Shlomchik, Warren D. Shlomchik
Abstract
Germline BRCA1/2 pathogenic variant (PV) carriers have elevated young-onset breast cancer risk. To define the pretreatment genomic landscapes of young-onset gBRCA-associated breast cancer, we evaluated 136 treatment-naïve tumors diagnosed before age 50 (92.6% ≤40): gBRCA1 86(63.2%); gBRCA2 50(36.8%) in the prospective POSH study, and 66 noncarriers from The Cancer Genome Atlas. Using whole exome sequencing, we analyzed somatic variation, allele-specific loss of heterozygosity (asLOH), homologous recombination deficiency (HRD), and single-base substitution signatures (SBS). gBRCA1(93%) and gBRCA2(96%) breast cancers had high rates of asLOH, but differed significantly in average HRD scores (57.4 ± 1.3 vs 43.7 ± 1.5, P < 0.0001) and median SBS composition (%): SBS1 (aging-associated) 12.9 vs 7.3, P = 0.013; SBS18 (reactive oxygen species [ROS]-associated) 1.4 vs 0, P = 0.007; and SBS3 (HRD-associated) 27.3 vs 42.6, P = 0.002. Compared to gBRCA2 tumors, gBRCA1 tumors with asLOH were significantly enriched for alterations in Hallmark ROS, DNA repair, and epithelial-mesenchymal transition pathways. In ER-positive, HER2-negative tumors from gBRCA1/2 carriers compared to noncarriers, we found significant enrichment of RB1 (OR:6.3;95%CI:2.8–15.4;padj = 0.001), TP53 (OR:4.6;95%CI:1.9–12.1;padj = 0.017), FAT1 (OR:3.9;95%CI:1.84–8.7;padj = 0.013), and MYC (OR:4.0;95%CI:1.8–9.1;padj = 0.017) SNV/indels/CNVs, associated with CDK4/6i resistance. Together, these findings demonstrate significant differences between gBRCA1 and gBRCA2-associated breast cancers, and preexisting CDK4/6i resistance mechanisms supporting prospective trials with individualized therapy for gBRCA1 vs gBRCA2 carriers, and comparing PARPi to CDK4/6i for ER-positive gBRCA1/2-associated breast cancer.
Authors
Mwangala P. Akamandisa, Mingyi Xia, Wilson Cheah, Bradley Wubbenhorst, Kurt P. D'Andrea, Mengyao Fan, Jake S. Shilan, Dana Pueschl, Anupma Nayak, Hayley McKenzie, William Tapper, Ellen R. Copson, Ramsey I. Cutress, Susan M. Domchek, Diana M. Eccles, Katherine L. Nathanson
Abstract
Background Cancer accounts for over 20% of late post-transplant mortality, yet the contribution of genetic susceptibility to post-transplant cancer risk remains unclear. This study investigates germline genetic risk factors for post-transplant cancer in the Finnish population using data from the FinnGen cohort. Methods A pan-cancer polygenic risk score (PRS) was constructed using genetic variants identified in UK and US populations to assess the influence of common germline variants on time to first cancer diagnosis in 1,802 Finnish kidney transplant recipients (KTRs), of whom 317 developed post-transplant cancer. The PRS was first validated in the FinnGen non-transplantation cohort and subsequently applied to KTRs, with replication in lung and liver transplant recipients (n = 476). Functional relevance was explored by assessing associations between the PRS and expression levels of 2,923 plasma proteins in the UK Biobank (n = 53,013). Results Compared to a matched non-transplantation cohort (n = 68,294), KTRs exhibited earlier cancer onset. The PRS was significantly associated with time to first cancer diagnosis in the non-transplantation population (HR 1.04; 95% CI 1.038-1.056; p = 3.75 x 10-25). Among KTRs younger than 40 years, higher PRS was associated with earlier cancer onset (HR, 1.08; 95% CI ,1.01-1.17; p = 0.036), indicating a stronger genetic effect at younger ages. The PRS significantly (Bonferroni < 0.05) altered the regulation of 87 plasma proteins, several of which were known cancer-related markers. Conclusion Inherited genetic predisposition, captured by pan-cancer PRS, may contribute to individual susceptibility to cancer after solid organ transplantation, particularly at younger ages.
Authors
Jarmo Ritari, Kati Hyvärinen, Kirsi Jahnukainen, Jukka Partanen, Ilkka Helanterä, Timo Jahnukainen
Abstract
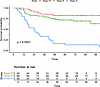
Merkel cell carcinoma (MCC) is a neuroendocrine carcinoma of the skin characterized by poor prognosis. This study aimed to explore the relationship between genetic alterations, tumor mutational burden (TMB), and MCC-specific survival (MCC-SS) in patients who underwent genomic profiling of tumors with OncoPanel. Univariate and multivariable analysis were used to assess the impact of genetic alterations on MCC-SS. Of the 188 identified patients, 164 were included in the analysis. The cohort had a mean age of 72.4 years (SD = 11.03), including 61.6% male. The median TMB was 5.32 (IQR = 3.04–25.53). Kaplan-Meier curves by high versus low TMB were significantly different (log-rank test, P = 0.017). PIK3CA (adjusted P = 0.003), SETBP1 (adjusted P = 0.002), KDR (adjusted P = 0.028), and RET (adjusted P = 0.033) were selected for multivariable analysis. In the multivariable regressions, only PIK3CA (HR = 2.07 [95% CI, 1.10–3.88]; P = 0.024) remained significant. PIK3CA remained significant across prespecified sensitivity analyses. In this study, high TMB and PIK3CA alterations were associated with poor MCC-SS. Identifying a higher-risk subgroup may inform risk stratification and motivate further evaluation of PI3K pathway targeting in future studies.
Authors
Matheus Lobo, Furkan Bahar, Julia L. Schnabel, Joao P. Duprat Neto, Aniket Shetty, Karam Khaddour, Manisha Thakuria, Ann W. Silk, James A. DeCaprio
No posts were found with this tag.